Duchenne Muscular Dystrophy Market Analysis by Size, Share, Growth, Trends and Forecast (2024–2032) | UnivDatos
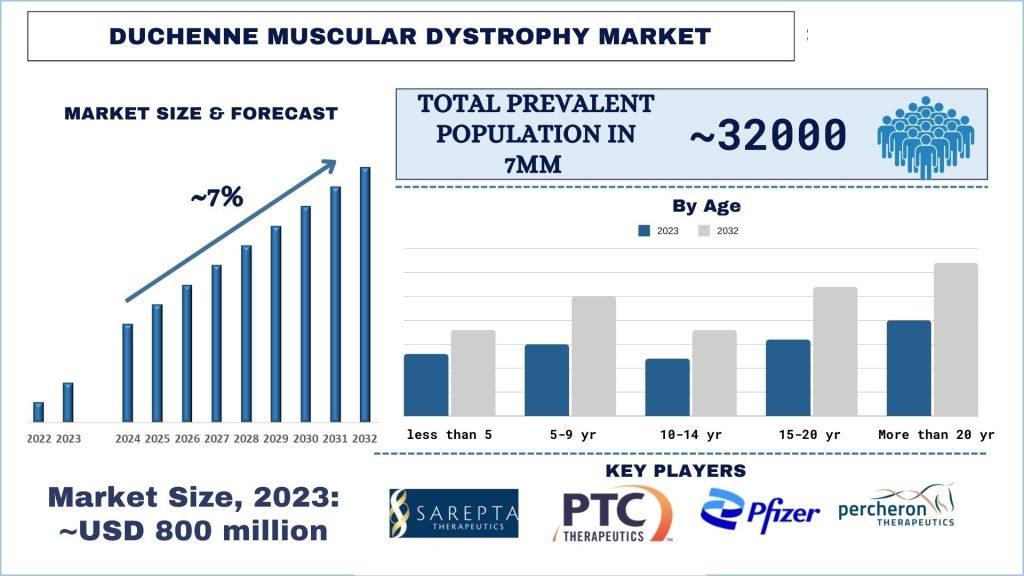
According to a new report by UnivDatos Market Insights, the Duchenne Muscular Dystrophy Market reached USD 800 million in 2023 and will grow at a CAGR of ~7% during the year 2024-32F. This is mainly because expanding treatment options will give patients access to a broader range of treatment options, enabling growth and penetration in the Duchenne Muscular Dystrophy industry as more therapies receive regulatory approval. For instance, on March 21, 2024, the U.S. Food and Drug Administration approved Duvyzat (givinostat) oral medication for treating Duchenne Muscular Dystrophy (DMD) in patients six and older. Duvyzat is the first nonsteroidal drug approved to treat patients with all genetic variants of DMD. This expansion will enhance personalized care approaches, allowing for better disease management based on individual patient needs.
Access sample report (including graphs, charts, and figures): https://univdatos.com/get-a-free-sample-form-php/?product_id=60948
Duchenne Muscular Dystrophy (DMD) is one of the most severe forms of muscular dystrophy, characterized by progressive muscle degeneration and weakness. Affecting primarily young boys, DMD leads to significant physical disability and reduced life expectancy. However, recent advancements in medical research have sparked hope, with emerging therapies poised to transform the DMD treatment landscape. This article explores the latest breakthroughs and trending therapies offering new possibilities for patients and their families.
The ability to tailor treatments to individual genetic profiles and specific disease manifestations enhances patient outcomes and drives market demand. Some of the key developments influencing the Duchenne Muscular Dystrophy market include:
· On March 28, 2024, Critical Path Institute’s (C-Path) Duchenne Regulatory Science Consortium (D-RSC) announced the launch of a groundbreaking model-based Clinical Trial Simulator (CTS) specifically designed to improve the design of efficacy studies for potential therapies for Duchenne muscular dystrophy (DMD). This pioneering Drug Development Tool is set to positively impact the medical research community by significantly optimizing clinical trial design.
· On January 5, 2023, Sarepta Therapeutics announced the signing of a commercial supply agreement for Catalent to manufacture delandistrogene moxeparvovec (SRP-9001), Sarepta’s most advanced gene therapy candidate for the treatment of Duchenne muscular dystrophy (DMD).
· On September 30, 2022, Solid Biosciences Inc., a life sciences company focused on advancing meaningful therapies for Duchenne muscular dystrophy (Duchenne), announced the closing of its acquisition of AavantiBio, Inc., a privately held gene therapy company focused on transforming the lives of patients with Friedreich’s ataxia and rare cardiomyopathies. The acquisition included its pipeline assets and net cash.
The regulatory framework's focus on personalized medicine has led to the approval of treatments tailored to individual needs and desired outcomes, enhancing patient satisfaction and driving market demand. Some of the most approved treatments for Duchenne Muscular Dystrophy include:
The treatment of Duchenne muscular dystrophy has included significant non-steroidal advances that offer new hope for managing the disease. Among these, Duvyzat (givinostat) stands out as a noteworthy development. Approved by the FDA in March 2024, Duvyzat is a non-steroidal histone deacetylase (HDAC) inhibitor that has demonstrated efficacy in reducing muscle deterioration across all genetic forms of Duchenne muscular dystrophy.
· On June 22, 2023, Sarepta Therapeutics announced U.S. Food and Drug Administration (FDA) accelerated approval of ELEVIDYS, an adeno-associated virus-based gene therapy for the treatment of ambulatory pediatric patients aged 4 through 5 years with Duchenne muscular dystrophy (DMD) with a confirmed mutation in the DMD gene.
Click here to view the Report Description & TOC : https://univdatos.com/report/duchenne-muscular-dystrophy-market/
Conclusion
In conclusion, the Duchenne Muscular Dystrophy (DMD) Market is poised for substantial growth, fueled by increasing prevalence, innovative therapies, and a supportive regulatory landscape. With a projected CAGR of ~7% from 2024 to 2032, the market registered USD 800 million in 2023. The market is witnessing significant advancements, such as the approval of Duvyzat and ELEVIDYS, marking a shift towards personalized medicine. Moreover, initiatives like the launch of the Clinical Trial Simulator and strategic agreements between companies further indicate a promising future for DMD treatment development and patient care
Related Healthcare Market Research Report
Sperm Bank Market: Current Analysis and Forecast (2024-2032)
India Catheter Market: Current Analysis and Forecast (2024-2032)
Blood Pressure Cuffs Market: Current Analysis and Forecast (2024-2032)
Next-generation Titrator Market: Current Analysis and Forecast (2024-2032)
Articulating Paper Forceps Market: Current Analysis and Forecast (2024-2032)
Contact Us:
UnivDatos Market Insights
Email - [email protected]
Contact Number - +1 9782263411
Website - https://univdatos.com/
Linkedin- https://www.linkedin.com/company/univ-datos-market-insight/mycompany/





